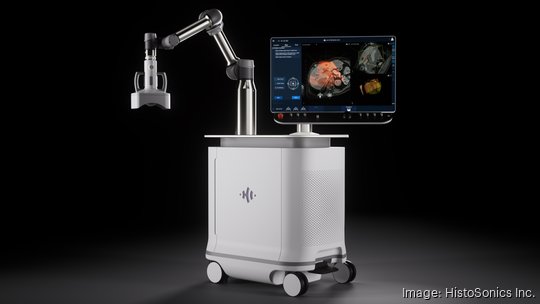
The Food and Drug Administration this month gave approval to HistoSonics Inc. to market the world's first histotripsy platform — a device that destroys cancerous tissue with sound waves.
The Plymouth-based company is now moving ahead quickly with commercialization of its Edison medical device, which is aimed at treating liver tumors.
It’s a landmark occasion for a company that was born at the University of Michigan in 2001 and officially launched in 2009.
“It’s an amazing milestone. Nothing that is this novel and new is easy when it comes to clinical trials and regulatory clearances,” said President and CEO Mike Blue.
For the first time, hospitals can begin to acquire the Edison system in routine clinical care instead of just as part of a more restrictive clinical trial. Edison was approved for liver tumors and HistoSonics has now begun a kidney tumor trial with plans to begin a pancreatic tumor trial early next year to eventually use Edison for those treatments also.
The market authorization happened through the FDA’s De Novo Classification Request process, a review pathway for medical devices that have no existing predicate. That means nothing like this exists so far.
“We began receiving purchase orders from hospitals across the country” immediately after the FDA offered De Novo classification, said Josh King, vice president of marketing at HistoSonics.
To Dr. Joe Amaral, vice president of medical affairs for HistoSonics, it’s rewarding to offer a procedure to destroy liver tumors without using a scalpel or needles. He said data and patient experiences in the studies gives the team confidence that histotripsy will have a meaningful impact on people suffering with liver disease and tumors.
The company has grown from about 40 employees a few years ago to 110, most of whom are stationed at the Plymouth headquarters. HistoSonics’ home base includes an education center where physicians from around the world have come for training on histotripsy.
HistoSonics charted one of the largest fundraising rounds last year, pulling in about $100 million near the end of the year. Across 10 funding rounds, the company has raised a total of nearly $227 million.
“Hopefully, we'll grow as the market allows us to," King said. "Our priority and our mission in the company is to offer meaningful change to patients and families suffering from serious disease."







