A device developed by Eagan-based Sonex Health will be used in a clinical trial to study its effectiveness for treating carpal tunnel syndrome against a traditional method.
Sonex Health, a medical technology company co-founded by former Mayo Clinic physicians Darryl Barnes and Jay Smith, and The Institute of Advanced Ultrasound Guided Procedures, which the two also founded, announced this week that the first patient has been enrolled in the U.S. clinical trial.
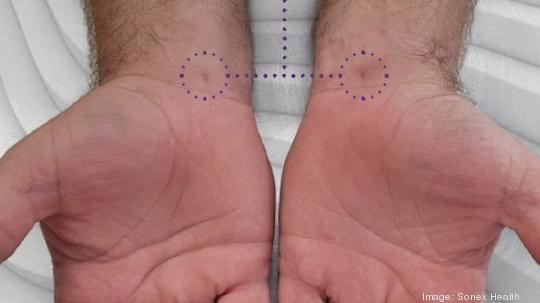
The prospective study measure Sonex’s single-use, handheld UltraGuideCTR device with real-time ultrasound guidance against conventional mini-open carpal tunnel release surgery.
The traditional procedure for carpal tunnel release includes making an incision at the base of a patient’s palm while real-time ultrasound guidance enables doctors to instead make a small incision at the wrist, the press release said. Sonex's device enables physicians to see all of the anatomy of the wrist and hand, allowing them to locate a "safe zone" and make space for the needed procedure, according to the company's website.
Founded in 2014, Sonex was launched out of the Mayo Clinic Business Accelerator program, the Business Journal reported. The company was also one of the first investments of Rochester-based Southeast Minnesota Capital Fund.
Sonex’s UltraGuideCTR received clearance from the FDA in 2016. The device went to market soon after and has been used on nearly 15,000 patients since then, said Sonex CEO Bob Paulson. Though he did not disclose how much capital has been raised since the company's founding, Paulson said the lead investor is Campbell, Calif.-based venture capital firm, KCK MedTech.
Data compiled from patients show Sonex's device allowed them to resume daily activities faster than the traditional method, Paulson said. A successful clinical trial will give greater assurances to insurance companies and physicians about its value as a treatment..
“I believe this technique is the biggest game changer in how carpal tunnel will be treated,” James Watt, an orthopaedic hand surgeon with Destin, Florida-based Orthopaedic Associates who enrolled the first patient in study, said in the press release.
The study will include a total of 120 patients and be conducted at nine sites around the U.S., including at Twin Cities Orthopedics in Minneapolis.
Sonex has also developed technology for the treatment of trigger finger, another type of repetitive stress injury. The device, called UltraGuideTFR, launched on the market earlier this year. The company is in the process of developing additional products, Paulson said.








