4C Medical Technologies Inc. announced Thursday it closed on a $35.4 million Series C round of funding.
The round was led by MicroPort CardioFlow Medtech Corp., a Shanghai-based medical device manufacturer.
In announcing the round, 4C Medical also said it appointed Sarvana Kumar as president and CEO.
Kumar has served as interim president and CEO since March 2021 after taking over for outgoing CEO Bob Thatcher.
The company is currently conducting a clinical research study for its device used to treat heart disease.
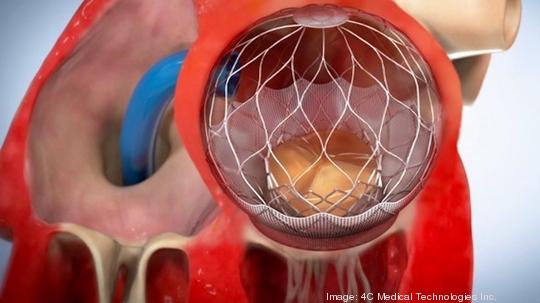
The AltaValve is a self-expanding stent that treats mitral valve regurgitation, a heart condition that mainly affects older people and causes blood to flow backward in the heart.
Other companies, including in Minnesota, are also working on devices that treat mitral valve regurgitation. What sets 4C apart is that the AltaValve is less invasive than other products on the market and preserves two parts of the heart, the native mitral valve and the left ventricle, that other products have to fixate on. That means the AltaValve is available to a much wider percentage of the patient population than other devices.
Kumar said in a statement the funding will allow the company to expand its product portfolio to include a device that is deployed in the tricuspid valve.
Founded in 2016, Maple Grove-based 4C has now raised $83 million. The company completed its $17 million Series A round in 2018; and followed that up with a $10 million round in 2020. It also raised around $8 million in 2019 and a $9 million seed round from Canadian angel investor Anges Québec and friends and family.