In our third annual Startups to Watch project, Triad Business Journal spotlights five local firms in growth mode, both in terms of their workforces and their reach.
They operate in vastly different sectors, from regenerative medicine to hiring technology to inventory solutions to health care. Yet, at their essence, they’re all driven by innovators focused on saving time, finding efficiencies and improving lives.
We started the rollout yesterday with a look at Beam Dynamics and continue this morning with Brinter, with profiles on Previewed, Three Strands Recovery Wear and Ventricle Health to follow.
BRINTER
Year founded: 2018
No. of employees: 14
Top executives: Tom Alapaattikoski, CEO and co-founder
Address: 391 Technology Way, Winston-Salem 27101
Website: https://www.brinter.com/
LinkedIn: https://www.linkedin.com/company/brinter/

Winston-Salem has seen numerous companies move their headquarters away, but, in 2023, one company moved nearly 4,500 miles to call Winston-Salem its home.
Originally from Turku, Finland, 3D bioprinting and regenerative medicine startup Brinter moved to Winston-Salem last year, with CEO and co-founder Tom Alapaattikoski saying that the city – which has a strong regenerative medicine presence – offered the most benefits to Brinter as a science company and as a growth startup. The company also has locations in Boston and Santa Monica, Califonia.
“We found a very thriving ecosystem and dedicated to supporting companies,” he said, adding that Brinter joined Winston Starts and partnered with the Wake Forest Institute for Regenerative Medicine through the RegenMed Hub Innovation Accelerator.
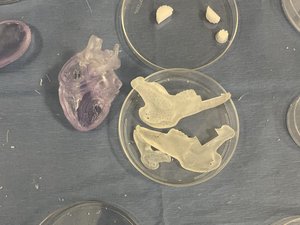
Brinter specializes in cartilage repair using implants made from 3D bioprinters, which it calls biomods. The company currently focuses on nose cartilage for surgeries such as facial reconstruction, rhinoplasty and nasal augmentation.
“Whenever you need more cartilage, you have to harvest it from other parts of your body – like from your ribs, for example. It’s a really painful, invasive surgery,” Alapaattikoski explained. “We’re going to replace that by manufacturing a cartilage material that mimics the native cartilage so we can have an endless amount of replacement cartilage.”
Not only does it use and become part of patient’s own cells, but Brinter’s 3D bioprinting technology allows for the implant to be personalized and unique to the patient.
With a goal of going commercial by mid-2026, Brinter has been focusing on hitting key milestones, Alapaattikoski said.
“We hit our first milestone, which is product development and materials development, a couple months ahead of schedule and we’re looking to start animal trials as soon as possible,” he said. The next big milestone will be to meet with the U.S. Food and Drug Administration, which Alapaattikoski hopes to do in the next few months.
Brinter is also raising a $2.25 million round that it will finalize by the end of Q1. When it was in Finland, the startup raised $4 million in non-dilutive grant funding, as well as about $2 million in seed funding.
In 2024, the startup also plans to hire for about three additional roles – specifically science- and business-focused – to grow its team in Winston-Salem. Alapaattikoski said the team will grow more rapidly as its product nears commercialization.
Alapaattikoski said that the company will also have big news coming in March, but he was unable to disclose what it is. In the meantime, Brinter will unveil a new website and bring in key advisors to help the company as it progresses towards commercialization.
“This is a really fast paced development we’re doing here to get our first product commercial in less than two and a half years,” he said.
The company’s technology will also have other applications for cartilage repair surgeries. For example, Brinter’s next biomod will be meniscuses for knee surgeries. Alapaattikoski expects that its meniscus biomods will be commercial by 2028 or 2029.
STARTUPS TO WATCH
- Beam Dynamics
- Previewed
- Three Strands Recovery Wear
- Friday afternoon: Ventricle Health