A local pharmaceutical startup is expanding its manufacturing operations to meet the growing demand of its first product, which launched in the fourth quarter of 2022.
Kannapolis-based Genixus, a pharma company that manufactures ready-to-administer syringes for medical staff to use in diverse hospital settings, is building a 30,000-square-foot production and distribution facility in Concord at 4715 Corporate Drive. Its existing 4,500-square-foot headquarters is in the main building at the David H. Murdock Research Institute in Kannapolis.
A Genixus spokesperson said the new facility is expected to open by mid-year. An investment cost was not disclosed.
To complement the increased production space, Genixus also plans to triple its staff by the end of 2024. It currently has just over 40 employees.
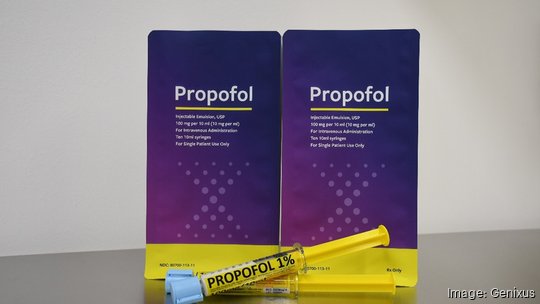
The expanded facility will provide ample room for heightened production of its initial product — ready-to-administer KinetiX RTA Propofol syringes. Propofol is a general anesthesia used in major surgery. The product launched after Genixus partnered with Kit Check, a national automation vendor in medication tracking. The company's syringes, which are available in 10- and 20-milliliter doses, are embedded with Kit Check’s Radio Frequency Identification technology.
Manufacturing of the Propofol syringes started at the company's Kannapolis headquarters and will expand into the new Concord facility, Genixus said. The augmented space will include formulation rooms for filling, inspection and packing, as well as quality control labs, isolators and automated syringe production.
Genixus' Propofol includes extended shelf life, ISMP compliant markers, color-coded plunger rods, an auto-dispensing cabinet and RFID-enabled doses.

"The finalized fit out, featuring new state-of-the-art syringe production and isolators, significantly expands our production and will play a pivotal role in addressing the demand for Propofol and our launch of additional KinetiX products in the second half of 2023,” said Kendall Foster, co-founder and CEO at Genixus.
Genixus, founded in 2019, announced in January 2022 the closure of $20 million in funding to bolster the development and commercialization of its product line. The startup is working to completely remove the prep work hospitals oversee for injectable medicine given to patients, the company previously told CBJ. Genixus ultimately hopes that will help eliminate manipulation, dilution and any chances for mistakes when issuing the medicine.
"We are passionate about simplifying injectable medications so clinicians can increase their focus on patient care,” said Seth Coombs, co-founder and chief commercial officer at Genixus. “The Concord manufacturing facility increases capacity for portfolio expansion within the KinetiX platform of injectables, allowing further solutions to be realized for the acute and critical care settings.”